Madeline Tompach, NIH Trainee 2021-2022
Madeline Tompach, NIH Trainee 2021-2022
Graduate Program: Molecular and Cellular Biology
Lab: Alicia Timme-Laragy
Research Interests: Investigating the impacts of preconception and embryonic exposure to per- and polyfluorinated substances (PFAS) on nutrient utilization and β cell development.
Research Summary
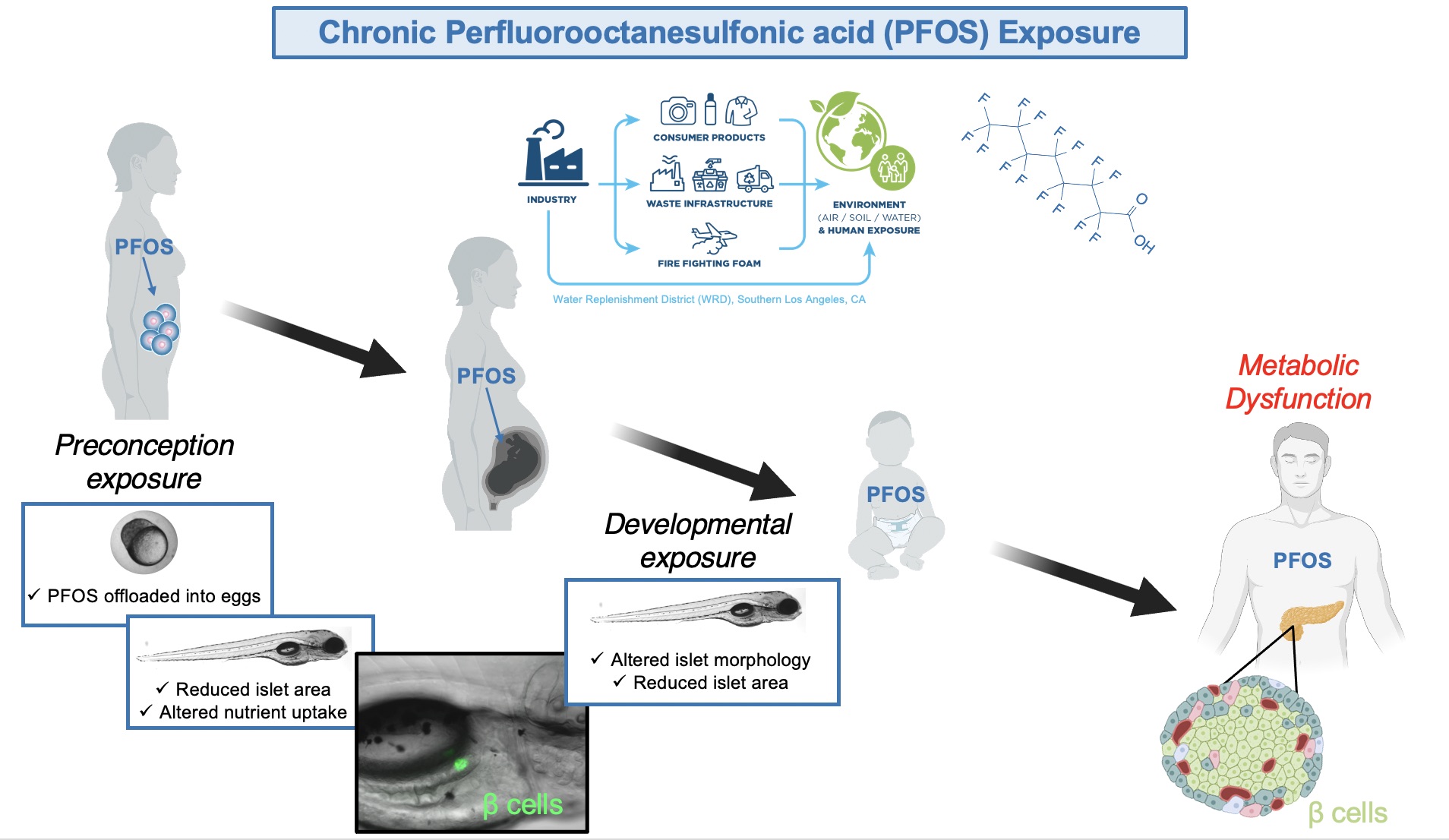
Per- and polyfluorinated substances (PFAS) are a large class of man-made organic compounds used as surfactants for a wide range of industrial practices and consumer goods. These chemicals have seeped into the environment where many are considered “forever chemicals” due to their high stability and resistance to degradation.
One such PFAS, Perfluorooctanesulfonic acid (PFOS), a component of nonstick products and firefighting foam, is found ubiquitously in the aquatic environment and drinking water supply. While the use of PFOS in the U.S. was banned in the early 2000s, its fully fluorinated, 8-carbon chain is not readily broken down in the environment or metabolized in living organisms, with a half-life of 5.4 years in human serum. Moreover, PFOS has been detected in >98% of human serum samples tested.
Humans are chronically exposed to PFOS beginning in development through placental transfer and breast milk. Chronic PFOS exposure increases the risk of metabolic disease in humans in part due the propensity of PFOS to target β cells, the insulin-producing cells of the pancreas. β cells are arranged into highly vascularized pancreatic islets with a rapid perfusion rate, increasing their susceptibility to toxicant exposure.
Our lab has shown that preconception and embryonic PFOS exposure in zebrafish leads to reduced islet size and abnormal islet morphology. Additionally, there is evidence that β cell function and mass are reduced with malnutrition during development, indicating a link between altered nutrient uptake and aberrant β cell development. Currently, we are studying altered nutrient uptake as a potential mechanism for altered β cell development with preconception and embryonic PFOS exposure. Additionally, we are investigating the antioxidant ⍺-lipoic acid (ALA) as a potential strategy to mitigate deleterious effects of preconception PFOS exposure on β cell health.